NHS England devastating U-turn on drug treatment for brain tumours
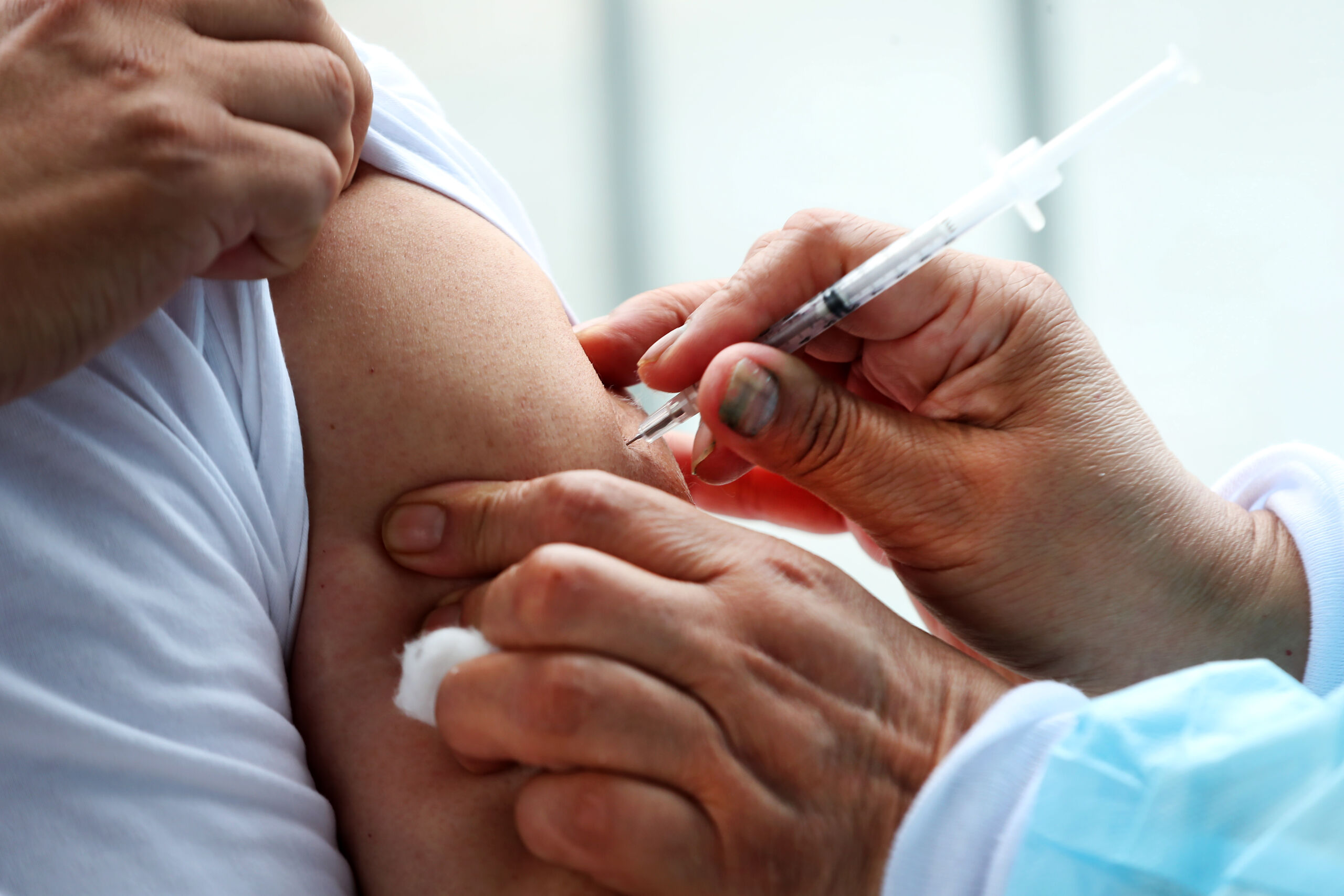
Last year NHS England promised to develop an in year policy for the use of Everolimus to treat SEGA (brain TSC related tumours). This month, NHS England
broke that promise and is saying this drug is ‘unaffordable’ for patients with inoperable SEGA. Each year we estimate that this will
affect 20 children and young people. It is absolutely critical that they have access to this medicine.
As way of comparison:
| The NHS has approved the use of Eculizumab for another rare disease, the most expensive drug in NHS history has been made freely available to all that need it. Costing a projected £82 million pounds to treat patients per year, the annual cost of prescribing this treatment is 200 times more then to provide Everolimus to TSC patients with inoperable SEGA. |
| It costs less to prescribe one person with Everolimus for a year, than it does to provide kidney dialysis for anyone experiencing kidney failure. Both treatments are potentially life-saving, but kidney dialysis is freely available on the NHS for anyone that needs it. |
This is deeply unfair and we must do all we can to protect the lives and health of young people with TSC.
Here’s why we hope you join us in being outraged at this latest decision:
- NHS England and the Health Minister George Freeman confirmed that a policy for routine funding would be published in the last financial year. That
promise has been broken. - NHS England delayed the decision on Everolimus for SEGA until June 2016, at which point they changed the way they make decisions on which treatments
they will fund. The new system massively disadvantages treatments for people with rare diseases. - NHS England have said Everolimus is ‘unaffordable’. There is no right of appeal or access to information on how that decision was made at the meeting.
- The Department of Health gets a rebate from the pharmaceutical industry of around £550 million pounds a year. This rebate is intended improve access
to new medicines. Although part of this money is used to fund the purchase of medicines in Scotland and Wales this does not happen in England.
We must not allow NHS England’s decision to go unchallenged. NHS England’s policy not only affects our community, but also that of other people with rare
diseases. We urgently need your help.
Here’s what you can do:
- Write to your MP{module_literature filter=”item” itemId=”140907″}
- Download the template letter and fill in the gaps, tell them why this decision needs to be changed.
- Find your local MP at www.theyworkforyou.com/mp
- Email them or post your letter.
- It is vital that we know which MP’s to follow up with on this issue, so please also either cc your email to social@tuberous-sclerosis.org or ring or email us to let us know what you’ve done.
- Share this story and campaign with your friends, colleagues and neighbours – we need as many people behind with us as possible. Share
our social media posts, and updates, and please use our #Fight4Treatment hashtag every time you do so. - Contact us if you think you or your child could be affected by this decision– we need case studies of to help us get news interest
in the story. Contact Emma Damian-Grint, Head of Fundraising & Comms, on emma.damian-grint@tuberous-sclerosis.org 0114 270 1723
If you are concerned that this decision will affect you or the person you care for with TSC, please contact your TSA Regional Adviser
PLEASE NOTE THAT THIS DECISION ONLY AFFECTS PEOPLE WITH INOPERABLE SEGA IN ENGLAND. THIS DOES NOT DIRECTLY AFFECT ANYONE IN WALES, SCOTLAND AND N.IRELAND.