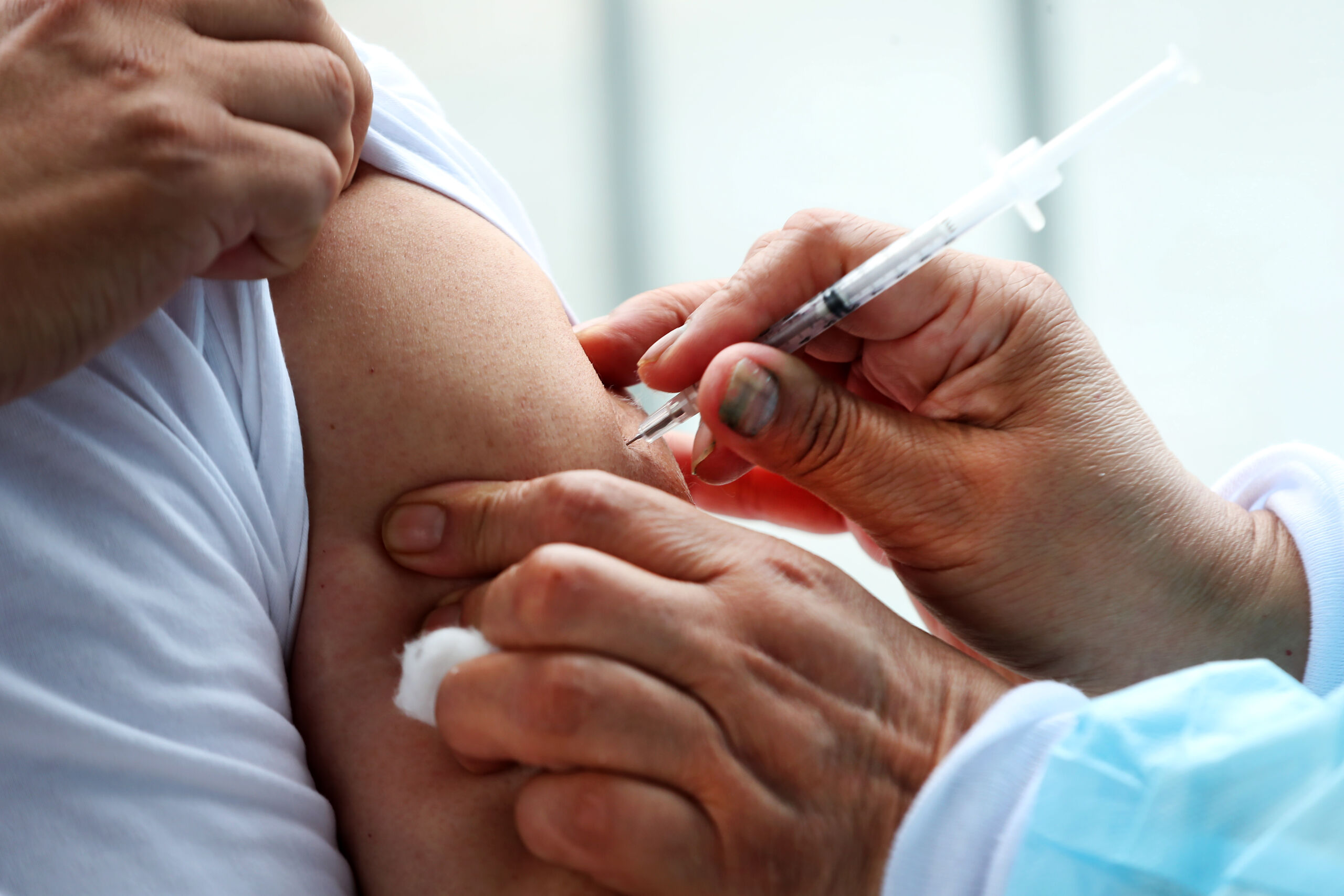
When Everolimus was licensed for the treatment of TSC, no-one imagined that more than 2 years later we would be campaigning for access in the face of a near total failure in NHS funding in England. Now, thanks to the many of you who have taken up the campaign and spoken out, we have achieved significant progress.
Two extremely important gains have been made:
Firstly, on 11th June NHSE published its policy on urgent and critical need. This means that any person who in the view of their doctor has a critically urgent need for treatment (those who cannot wait for a national policy to be developed) should now get NHS funding. The new policy will benefit all patients with critical need where there is no national policy – in addition to those with TSC.
Secondly, in a communication to the NHS issued on 2 July, NHSE confirmed the development of national policies for the prescribing Everolimus for TSC (SEGA and AML). Development of these policies has been speeded up and a decision is expected in the autumn. If agreed, funding will be made available immediately – and Everolimus for TSC will be routinely available on the NHS before the end of this year.
A massive “thank you” to all who have taken part in the campaign so far – without you, these important gains could not have been made.
Firstly, on 11th June NHSE published its policy on urgent and critical need. This means that any person who in the view of their doctor has a critically urgent need for treatment (those who cannot wait for a national policy to be developed) should now get NHS funding. The new policy will benefit all patients with critical need where there is no national policy – in addition to those with TSC.
Secondly, in a communication to the NHS issued on 2 July, NHSE confirmed the development of national policies for the prescribing Everolimus for TSC (SEGA and AML). Development of these policies has been speeded up and a decision is expected in the autumn. If agreed, funding will be made available immediately – and Everolimus for TSC will be routinely available on the NHS before the end of this year.
A massive “thank you” to all who have taken part in the campaign so far – without you, these important gains could not have been made.